The Rate of Reactions
HTML-код
- Опубликовано: 23 июл 2024
- 035 - The Rate of Reactions
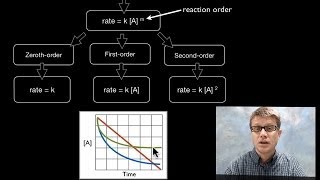
In this video Paul Andersen defines the rate of a reaction as the number of reactants that are consumed during a given period of time. The rate of the reaction can be affected by the type of reaction as well as the concentration, pressure, temperature and surface area.
Do you speak another language? Help me translate my videos:
www.bozemanscience.com/transla...
Music Attribution
Title: String Theory
Artist: Herman Jolly
sunsetvalley.bandcamp.com/trac...
All of the images are licensed under creative commons and public domain licensing:
"Beer's Law Lab." PhET. Accessed September 23, 2013. phet.colorado.edu/en/simulatio....
"File:Handwarmers.JPG." Wikipedia, the Free Encyclopedia. Accessed September 23, 2013. en.wikipedia.org/wiki/File:Han....
"File:Large Bonfire.jpg." Wikipedia, the Free Encyclopedia, September 22, 2013. en.wikipedia.org/w/index.php?t....
"File:Rust.rost.JPG." Wikipedia, the Free Encyclopedia. Accessed September 23, 2013. en.wikipedia.org/wiki/File:Rus....
"File:Rust03102006.JPG." Wikipedia, the Free Encyclopedia. Accessed September 23, 2013. en.wikipedia.org/wiki/File:Rus....
"Reactions & Rates." PhET. Accessed September 23, 2013. phet.colorado.edu/en/simulatio....








You're just awesome for taking the time out of your day to help out with amazing videos
Thank you for doing what you do
just saying he gets ad revenue tho it might be small
This guy explained in 6 minutes what my teacher failed to do in a day
And to honest, the whole semester
Y (same)
lmao 🤣
My teacher sent us this video
bro why the hell do our teacher fail to teach like this like it's honestly such a big issue you see comments like these on all these youtube channels like Khan academy and also OCHEM tutor like wth
You da Real MVP. These videos are helping me in my general chemistry II class in college.
Da hell we doing this semester 4 junior year????
Where has this channel been all semester? It all makes sense. Thanks. Great video.
Animation really helps in explaining facts and figures more clearly.Thanks.
Always thankful for your efforts!
Give this man a Nobel peace prize
Peace is not an award but a responsibility
As an online student, I don't get the lecture...if I'm lucky I get a 4 minute video from the professor. Your videos have helped me in my Chem, Bio, and Physics classes. It is because of you and your ability to explain complex topics in simple terms that I have been able to pass these classes while also understanding the topics. Thank you a million times over!
Not even learning this, but Mr. Anderson just makes me remember. Thanks for doing what you are doing!
MY teacher is so self centered that when he is not talking about himself he is judging us for asking stupid questions. I love chemistry but he makes it so complicated. Thank you so much. U saved my grades.
Mr. Andersen thank you so much for posting these videos online! I watch your videos supplementary to my biology lecture and I can honestly say that they help me transfer the information from my short term to long term memory. I know I'm coming back here to review for midterms! You're greatly appreciated here(:
Thankyou so much! I was lost without this!
As amazingly helpful as always
Excellent lecture❗
*Thank You*
that was helpful! but how would i answer this question? it says to state the easiest way to measure reaction rate and in the question, there is an equation with different colors written to each of the reactant and product!
Thank you good sir.
atlast anderson sir is back with a new video .
Thank you!
thank you for your video
thanks 4 your effort it was very helpful that you hope and i want you to make a video about nano technology and some applications on it like medical ones and explain how the"-cooh"carboxyl group is an acid although it has oh-
Thankyou Sir it really helped me a lot.
Me too. I aced chemistry because of this guy.
Maybe should have added some rate of reaction problems?
Hello, mr.Andersen, I just wanna know what is that simulation program you are using in this video?
Thanks, it is a great video.
phet.colorado.edu/en/simulation/reactions-and-rates I think
THANK YOU YOU ARE A MILLION TIME BETTER THAN MY TEACHER!!!
+Mohammad Hussain lol
@Montaga wtf its really been 4 years, im in uni now
@@MuhammadHussain-ry9vh Wow
@@yasmeenaraedeldilie1278 Damn its really been 5 years
Thank you.
thank you
Hey, thanks for your videos. Do you have a link for those interactive demonstration animations?
This is sometimes what some teachers fail to teach---the practical application of science.They can explain very well the topics in a molecular level but their students sometimes end up questioning themselves 'What the hell is that for?'.
I am already in college, taking up BS MedTech. Your videos really are a great help.
belated happy Teachers' Day! -----ivhie from The Philippines
You are amazing
my nigga bozeman
What are you using to demonstrate this and can i download it/use it?
spaciousinvader1 Phet simulations :)
very much helpfull.
U R A LEGEND ....
thank you soooo soooo much I got it now I wish if my lecturer explaining clearly like you
thank u
Thx!
You are genius thank you so much sir
thanks!
the site that you do in them experiment what there name plz ?
Taha Daboussi phet simulations
Great
I have question . plz reply
An enzyme increase the rate of reaction by an order of 10^10- 10^15 times as compared to an uncatalyzed reaction. If an enzyme takes one sec for a particular reaction, what is the minimum required for such a reaction in its absence? ( ans should be in years)???
A Catalyst will not change the amount of product obtained
Good
Keep doing
nice
good video but I'm still confused
Wow
what about catalyst?
he forgot that alright
WASD or IJKL or ESDF
today my teacher explain for 3 hours only about rate law and rate constant. not his fault though. it's mine
Always learn with mr boze. And he's cute!!!!!
yeah, my teacher is not organised. the white board is such a mess. when he write, he show his ass. i don't see what he wrote. then, he blame me for not focusing and good at fundementals. not his fault though. some people born the way they are
This comment was an adventure
God make me some smart chemist......
Hello.
mmmmmmmmmmmm crunch
either too easy or im missing out something BIG
A chemical catalyst is also a factor
+Peter Arnold true
Yup
suraj
ponz
Fattthhheeeeeerrrrrrr
ty noob
Maybe should have added some rate of reaction problems?
thank you
thank you