Naming Coordination Compounds - Chemistry
HTML-код
- Опубликовано: 14 янв 2018
- This chemistry video tutorial provides a basic introduction into naming coordination compounds. It contains plenty of examples and practice problems on the nomenclature of coordination compounds. It discusses how to determine the oxidation state of the transition metal cation and how to name it when the complex ion is cation and when it's the anion.
How To Balance Nuclear Equations:
• How To Balance Nuclear...
Alpha, Beta, & Gamma Decay:
• Alpha Decay, Beta Deca...
Half Life Chemistry Problems:
• Half Life Chemistry Pr...
Carbon-14 Dating Problems:
• Carbon 14 Dating Probl...
___________________________________
Nuclear Binding Energy & Mass Defect:
• Nuclear Binding Energy...
Nuclear Chemistry & Radioactive Decay:
• Nuclear Chemistry & Ra...
General Chemistry 2 Final Exam Review:
• General Chemistry 2 Re...
SAT Chemistry Subject Test Review:
• SAT Chemistry Subject ...
____________________________________
Coordinate Covalent Bond:
• What is a Coordinate C...
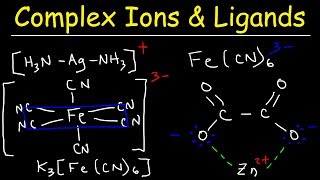
Complex Ions & Ligands:
• Complex Ions, Ligands,...
Naming Coordination Compounds:
• Naming Coordination Co...
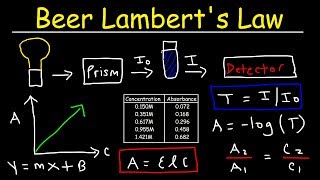
Beer Lambert's Law:
• Beer Lambert's Law, Ab...
Crystal Field Theory:
• Crystal Field Theory
___________________________________
ACT Math Practice Test:
• ACT Math Test Prep
Final Exams and Video Playlists:
www.video-tutor.net/
Full-Length Videos and Worksheets:
/ collections
Chemistry PDF Worksheets:
www.video-tutor.net/chemistry...






Next Video: ruclips.net/video/rllHziqWlgU/видео.html
Chemistry PDF Worksheets: www.video-tutor.net/chemistry-basic-introduction.html
Final Exams and Video Playlists: www.video-tutor.net/
When will I be able to see the face of the man who has helped me succeed in my upper science classes? : )
I'm sure his face will be as pleasant as his voice.
Soon he hits 10M subs then
You have a crush on him don't you
nair u don’t??
@@nair7961 who doesnt?
For anyone confused on why we use latin endings for some of these...If the ligand part is a CATION we use regular names like Cobalt. However if its an ANION we use the latin name...Colbaltate.
Not the Latin name we add an ate to the Latin name eg : argentium argentate
Sir u are almost saving my 2 classes physics ,chemistry and mathematics too. Thank you from the core of my heart you are a life saver 😃😃😉😉🤗🤗
Which country?
India
"feel free to pause the video"... "so for those of you have solved it congratulations, you are an excellent subscriber"
We need more educational videos like this on youtube. This is the future of education. Thank you "The Organic Chemistry Tutor"!!
hey ! i am from india in our education system this topic is in 12th chemistry, at what level you are taught this topic ? just wanna know
This guy deserves more than half of my school fees. good job bro , I would like to see you. Share with your fans your social media names.
Thank you so much for this hospitality towards your learning students. I do belong from Pakistan and the way you teach, so there is no such exaggeration in the assertion that you're one of the greatest teacher, i have ever seen. Thanks a-lot :D
agreed
Me too watching from Pakistan
Most comprehensive video ever made. THANK YOU SO MUCH. If all lectures were like this literally everyone would graduate with a distinction
I totally appreciate this man for helping me out in understandingVarious concepts of Physics chemistry and math.The derivations which were difficult to understand in my physics text book he made them easier to understand.I appreciate this man for sharing his wonderful knowledge with us!
it is been already 3 years since I discovered your channel I passed physics, chemistry and mathematics which I really hate was the only subject that I got an A+ so thank you so much you are a hero. here I am in having a chemistry exam tomorrow and watching your videos
You are saving many lives.. You are a great teacher!.. thankyou for your wonderful tutorials 💖
This is so simple than it appeared. I just had to subscribe.
Thank you.
Hoping to improve using this channel
Thank you so much you made this subject make so much sense to me and now I enjoy it!
I don't know whether you- the creator of this video- will read this comment or not but let me just say this: Thank you for uploading this! You've been so helpful. From the bottom of my heart, thank you!!😊😊
It’s really a wonderful video to learn nomenclature of co-ordination compounds
Kind sir, thank you so much we appreciate everything you do! I hope you feel like a good person and you're having a great day
I don't know how to thank you. You have helped me a lot.
your voice is calm and the way your teach is so perfect i always watch your videos on differents subjects and they helps me too, finally i really appreciate your works hard. thank you again.
really love how your videos are my saving grace in medical school
I'd be lost without you, thanks for existing
my major is chemistry and I almost graduate after 3 years in chem my gratefulness and thanks will be all for you, I might miss chemistry but I'll miss your videos more, THANK YOU, SIR :')
Amazing explained sir, i don't know why you tube recommend this channel after a long time, but you are awesome, thanks 🤗
Love from India 🇮🇳
Thank you so much u have really help me right from my secondary school to my uni
May God continue to bless you
Thanks bro, that one there was a nice video.
Eventhough it was a small portion it was amazing and it helped me to understand very well..
Relaxing voice. Ahan😊 i like it!
Man you are awsome! honestly i watched everyone of your organic videos and now i learned it so well that i can remember it on my fingertips,all thanks to you.Keep up the good work you have a bright future ahead of you.
The most difficult topic has become the most interesting to me ...
Just because of the great teacher .... Thank you from the core of my heart.... ♥️🙏
Wish you the best of the best
Thanks for everything you did for us
You always helped me in highschool now you are helping me in pharmacy college .❤️🖤
SO DIRECT AND UNDERSTABLE. BEST ONE!!
You really are a life saver. I can't thank you enough.
Literally watching just before my exam....thamks ❤❤❤
Every time I watch your video, I'd click like even before the video starts because I know the content is gonna be really helpful and easy to understand. You're really the hero of my study, thank you so much.
You are amazing, thanks for clearing the concepts!
I'll forever be grateful for these lectures that are truly amazing and facile...tqsm♥
You got the BEST video on this topic!!!
Finally seems like I've learnt this, thank you so much!!
Wow! You are a fantastic chemistry teacher!
Saving my life at this point again
How privilege we are to av such an amazing teacher thank u sooo much sir👏🌹
It was simple clear and super easy thanks a million ♥️♥️♥️
ZabarDAST video!!!! Sab kuch samajh aa gia ha...clapping....
To the point explanation. Good job 💯 points for you mister 😊😊
Thank you so much for making these videos!!
Yes it gave me good understanding. Thank you I was studying for exam.
Small thing I noticed in the video as that you're using older naming conventions. The current IUPAC-recommendation is to call Cl- ligands "chlorido" instead of "chloro", Cn- ligands "cyanido" instead of "cyano", and the same with other ligands ending in "-ide" or "-te". Lots of older sources will still use the old nomenclature but this is technically incorrect.
Oksalayo!!
Agreed
Thanks for letting us know. Useful
I literally commented that and saw urs lmao.
Tnk u sir....easy understanding with clear voice.....😃😃
Very helpful......I understood it very easily
You are so good man. I understood it in one go.
Your videos are always great. Thanks
If i was asked for a wish then i would wish to meet u sir becoz there is no topic left unlearned and u upload all the vedios wich i needed ..
number of ligands, then ligand name (place ligands in alphabeitcal order), transition metal, transition metal's oxidation state as the number, then anion
Excellent explanation thank you!!!
Thank u so much.. Its really a wonderful lecture
Thank you sir, i'm all the way from South Africa and you're my life saver i don't wanna lie , thanks once again😃😃🥵🥰
Complete all concept in 10 min . Great sir 👌 love from Pakistan 🇵🇰♥️
This videos are so helpful 😭😭😭i really appreciate you
Saving lives since 1980. Thank you Sir
Man what are you you make my life so easy thank you so much
Omg thank you so much you are a life saver 😫😫😫😫♥️♥️🙏🙏🙏
Outstanding sir thank you very much to clear concept
sir thank you for the video! i came to discover something that when there is anion complex the metal ion name would be written as cobaltate ( ate ) but when its cation complex, the metal ion would be written as the original name of the element. like cobalt. ^_^
Life is now simple because of you!!
Hi, I would just like to ask if we could also use chlorido instead of chloro?
this is my fav channel
Thank you very much sir. This video has really helped me
watch this ruclips.net/video/brtQPfwUOt0/видео.html
Thank u so much sir you done a good job🙏
Thank you sooooooooo much sound plus knowledge plus ur calmness zzzzzz. Like em
you are a LIFESAVER!
thank u 15 min over 3 months
Nice Video that prvides mostly correct information. However, a complex (coordination compound) has to be wrtitten in square brackets, you're often forgetting that. And BTW, I never heard of hexachloroplatinate(II), but hexachloroplatinate(IV)
Thankx a lot it for explaining it so well ... ❣️
Would have failed chem 2 if not for these videos, honestly thank you!!!!!
Nice explanation.thank u very much.Johnson-India
for iupac standards aren't ligand anions like chloro written as chlorido and aren't the latin names disregarded so you would still put down Iron(III) rather than ferrate
Very good explanation ............ thanks
Thank you so much for your guidances
thnakx bro .you have learn me how to name co.comp.Thanks.
Thanks a lot sir...You made this easy....
BLESS YOUR SOULLLLLLLLLLLL THANK YOUUUUUUUUU
It helped me so much thanks a lot❤
Awesome! Thank you! The most awaited video!
Amrutha.
I'm from Brazil and this video really helped me out. I just had to make some adaptations but it was almost the same. thank you very much.
Thank you sir it helps me a lot ...
Thanks sir...... Video is really very helpful.....!
Thank you sirrrrr for saving mee❤❤❤god bless you 🙏❤
Thanks cause you make me understand better
I'm gonna cry. I just, wanna thank you. You made it sooooooo much easy. God bless you.
Thank you sir. It was very helpful... your explanation was really wonderful... 👍👌
geetha watch this one ruclips.net/video/brtQPfwUOt0/видео.html
You dropped this, King 👑
this man is breathtaking
Sir in the second compound shouldn't the chloride part be chlorido instead of chloro since the system has changed?
So shouldn't the compound be
pentaammine chlorido cobalt (III) chloride?
Just a question, how did you get the charge on CN to be -1?
Sr. When halides eg chlorine comes inside the coordination entity is it named as chlrido?
Thankss. Helped me so much.. ❤️❤️
Very very useful...thank you sir.
Thanks alot, may God Bless you
Thank you so much !!!!!! You've helped me understand it so clearly!! ❤️❤️❤️
Very helpful thanks!
In the second example, why is the chlorine inside the cation complex not considered for calculating oxidation state of the central metal atom?
I am from Iraq, and I enjoyed your explanation of finding the oxidation number of the central atom of the coordination complex