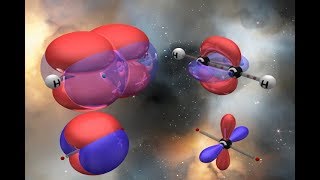
Hybridization of H2O (water)
HTML-код
- Опубликовано: 17 окт 2024
- First, hydrogen doesn't hybridize.
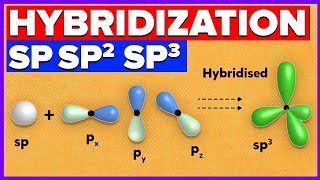
The oxygen DOES though, to explain the two sigma bonds PLUS the two lone pairs. It requires FOUR hybrid orbitals and so the hybridization is sp3
Check me out: www.chemistnate...







This is the simplest explanation I've come across... Big thanks
easy and simple that deserves a like for sure !
So helpful ! Thank you!
just exactly how SiO2 is sp³ geometrically?
I ain't kid but thank you 😭
Thanks I m from India I seen your chemistry 🎉
helpfull
Helpful