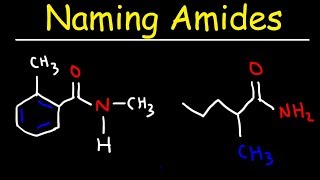
Naming Amides Using IUPAC Nomenclature for Organic Chemistry
HTML-код
- Опубликовано: 3 окт 2024
- leah4sci.com/or... presents: Naming Amides
This video is Part 19 in the Naming Organic Compounds series
Are you struggling with organic chemistry? Download my free ebook "10 Secrets To Acing Organic Chemistry" here: leah4sci.com/or...
This nomenclature tutorial video takes you through the IUPAC rules for naming amides including simple and substituted amides, as well as cyclic and diamides
Many students struggle with naming because they attempt to come up with the entire name at once and often wind up missing a piece or two. After this series you won't have that problem anymore
Visit my website for additional naming tutorials:
leah4sci.com/naming-organic-compounds-iupac-nomenclature/
Information on scheduling a tutoring session with me: leah4sci.com/or...
And of course my ebook: "10 Secrets To Acing Organic Chemistry" forms.aweber.co...








I wish I would have found you when I took Ochem 1! I'm at the end of Ochem 2 and I have learned so much with your videos in the last few hours than in days figuring it out on my own!
Thank you!
You're very welcome, so happy I could help!
You are simply amazing. One of the best tutorial videos I have seen online. Bravo
Please add videos about common names too
thanks so much for your kind words and your suggestion!
Thanks heaps, your videos are very helpful. You're a great teacher - very clear, concise & simple in your methods. I'm taking university preparation chemistry & your videos have been a big help! :)
You're so very welcome, I'm glad I could help!
Thanks a lot..Got IUPAC nomenclature test tomorrow!
You're welcome! I hope your test went well
Leah4sci it did! Scored 96/120.. Second in class.. got messed up with priority order
thank u very much.God will bless u for helpin us
You're very welcome!
Perfectly explained - great video!
Glad it helped!
thank you!!!!!!!!!!!!!!!!!!!!!!!!!! Doing my best in Ochem, but it does not seem to be enough. Your video was to the point and not "sleepy" like some of the rest of them. I was captured. It was organized. Thanks!
So glad I could help you without putting you to sleep lol!
Thanku it was simply amazing 👍
You're welcome!
Thank very much,you share a lot of pearls of wisdom...This is very helpful..
you're very much welcome Junnik :)
CLEAR EXPLANATION. LIKE IT
Glad you liked it!
This is great and straight forward :)
Glad you liked it!
Thank you very much! I am from India and I am preparing for IIt-Jee (if u know about this exam). You helped a lot.
You're welcome!
Thanks Leah
I have a question. Do you know how to name amides by their common names?
I'm sorry, but I don't offer tutoring through RUclips comments. For help with this and more, I recommend joining the organic chemistry study hall. Full details: leah4sci.com/join
this is really good I look forward to such videos
Glad you liked it!
very concise. thank you.
You're very welcome!
The basic concept of chemistry is so curious to understand from ur videos..
I want more mechanism type videos
Since i study organic better
I love organic
My lover is organic
Happy to help!
than you soo much :'> a really great help for my report tom.
You're very welcome, happy to help!
You are amazing thanks you so much
You're welcome 😊
0:47 NH2 not NR2
You are the best 💕
Thank you!
excellent video. thank you so much!
You're very welcome!
how funny you're answering me now, 10 yrs later. better than never, i guess. lol@@Leah4sci
Why you should not show the position of amide in suffix?
This is because an amide is always terminal, and as the highest priority functional group, it will always fall on Carbon 1. It is NOT possible for an amide to be within the chain.
Thank you so very much!💯
You're welcome! :)
thank you so much!!
You're so very welcome!
Thank you so much!!
you're welcome!
does the number ALWAYS COMES FIRST in naming if there is 'N' in naming amides or amines?
What number are you referring to?
1:49, what if it had a H3C in carbon 4?
Adding a methyl group to the terminal carbon would lengthen the parent chain by one additional carbon. Rather than "butanamide", the parent name would be "pentanamide" because of a 5 carbon chain.
Nice video very helpful
happy to help!
Benzene or benzamide more reactive?
Depends on the reaction
Great video
Glad you liked it!
Thanks mam
you're welcome
Thank
welcome!
Thank you so much!! Godblessyou!!
You're very welcome!
Good gob
Thank you!
fantastic :)
So glad you liked it!
❤
Thanks for watching!
go slow it's fast for us to understand
Have you tried watching the video at a slower speed?