21.1 Introduction to Coordination Chemistry | General Chemistry
HTML-код
- Опубликовано: 21 июл 2024
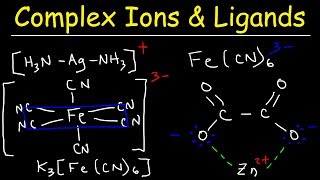
- Chad provides an introduction to Coordination Chemistry in this lesson. The topic revolves around the coordinate covalent bond which is formed between a central metal ion acting as a Lewis acid and one or more ligands acting as Lewis bases in the formation of what are called complex ions and coordination compounds. A list of the most common ligands is presented. The coordination number is the number of bonds made to the central metal ion, and the most common coordination numbers of 2, 4 and 6 are discussed. For a coordination number of 2 the geometry is linear. For a coordination number of 4 there are two possible geometries: square planar and tetrahedral. For a coordination number of 6 the geometry is octahedral. Polydentate ligands are also discussed which can form more than one bond to a central metal ion. They are also commonly referred to as chelating agents, and complexes formed between metal ions and polydentate ligands are often referred to as metal chelates.
I've embedded this playlist as a course on my website with all the lessons organized by chapter in a collapsible menu and much of the content from the study guide included on the page. Check this lesson out at www.chadsprep.com/chads-gener...
If you want all my study guides, quizzes, final exam reviews, and practice exams, check out my General Chemistry Master Course (free trial available) at www.chadsprep.com/genchem-you...
00:00 Lesson Introduction
00:32 Coordinate Covalent Bond
02:28 Coordination Number and Geometry of Complex Ions
03:47 Coordination Sphere
07:51 Polydentate Ligands
www.chadsprep.com/
courses.chadsprep.com/pages/p...



![Sean Rii, Karyon, Sharzkii - Taungule [My Love] (Official Music Video)](http://i.ytimg.com/vi/eJoMuypbSzQ/mqdefault.jpg)




Using this to study for my inorganic chemistry final - this is going to be a life saver.
Hope you do well on your final - Happy Studying!
Very nice, Chad. I might actually pass tomorrow's test.
Thanks - I hope you do well.
Do you happen to have a video on 18 electron rule with the ionic method and covalent method?
Your voice is actually so soothing! Helped me study so much!
Glad it is - Happy Studying!
Hi Chad! I finally got into dental school (I used your studies) and now I'm taking chemistry in dental school and I am still watching your videos to review and learn things I forgot overtime. I thank you with all my heart!
Congratulations Hazel! I hope you had a little bit a time to just feel good about where you've gotten, before getting after it again!😊 Best wishes to you in dental school and beyond!
You made inorganic interesting to me....thank you very much
Most welcome!
this was extremely useful 🤸🏽🤸🏽
Excellent!
Good day sir, i watched you a lot. could you upload more in coordination chemistry. Like structural and optical isomerism
Several more lessons on this chapter coming throughout the week. Naming complex ions/coordination compounds later this morning and a detailed lesson on isomers tomorrow! Hope this helps!
your lectures are so good
Thanks for saying so - Happy Studying!
Thank you for all your helpful videos. I think that in the formula of EDTA 4 C atoms are missing...
You're welcome :) is this in the chart around 10mins?
what's the charge on the Fe in [Fe(H2O)5 Cl] Cl2, is it Iron +2 or Fe +3 ?
+3 to balance out the Cl ligand and the 2 Cl counter ions. 😊
thx u helped me alot on my tests
Glad to hear that!
Perfect 💛
Thank you :)
loved it!
Excellent!
Really helpful 👍
Excellent!
Why do we have 2+
God bless u
Thank you - to you as well.
Nice sir! You looking is too young
Thanks!
Speak slowly you are teaching some Africans.We can't understand you sir.If you can speak slowly many of us will always view your video and subscribe.
You can change the playback speed under the settings to something more manageable. Happy Studying!
You are talking so fast
I get that a lot - you can change the playback speed in the settings. Hope that helps.
@@ChadsPrep thank u❤
Happy Studying!