The Rate-Limiting Step
HTML-код
- Опубликовано: 9 ноя 2013
- 042 - The Rate Limiting Step
In this video Paul Andersen explains why the slowest elementary step in a chemical reaction is the rate-limiting step. This step can be used to determine the overall rate law of the chemical reaction.
Do you speak another language? Help me translate my videos:
www.bozemanscience.com/transla...
Music Attribution
Title: String Theory
Artist: Herman Jolly
sunsetvalley.bandcamp.com/trac...
All of the images are licensed under creative commons and public domain licensing:
File:Carbon-monoxide-3D-vdW.png, n.d. commons.wikimedia.org/wiki/Fil....
File:Nitric-oxide-3D-vdW.png, n.d. commons.wikimedia.org/wiki/Fil....
"File:Nitrogen-dioxide-3D-vdW.png." Wikipedia, the Free Encyclopedia. Accessed November 2, 2013. en.wikipedia.org/wiki/File:Nit....
"File:Oxygen Molecule.png." Wikipedia, the Free Encyclopedia. Accessed November 2, 2013. en.wikipedia.org/wiki/File:Oxy....
Jynto. Space-filling Model of the Carbon Dioxide Molecule, One of the Most Important Chemical Compounds in the World - Vital for Life as We Know It, but Catastrophic at Excess Levels., July 15, 2011. Own work. commons.wikimedia.org/wiki/Fil....


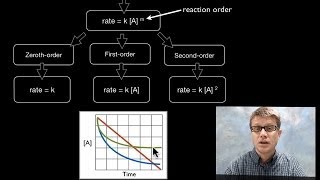






I still don't understand just by looking at the equations how you know which one is going to be the slow step. Is it always the one that's not reversible? I've come across many AP practice questions that ask you to determine which is which and I have no idea how.
Wow....you sir have a talent....I don't think I will ever forget this analogy...
I have a question. we know that rate determining step is the slowest part (step) of the mechanism but how do we by looking at the equations which is slow and which one is not? this is the main part of the whole thing. can anyone help!
4... years... later
hi im 5 years late to tell you that you will be given more information in a quiz like the activation energy for each step so you can tell which step is the slowest. hope it helps someone lol.
no
@@monxampion6930 thats good to know thanks
@@monxampion6930 thank you I was really confused about that!
Such a fantastic person and a teacher. An absolute blessing.
Awesome analogy - thanks for the explanation!
Finally a video in plain english ! thank you so much!!
this is fantastic, great analogy! Really understand this now thank you
Rate is determined experimentally. One can't look at the number of atoms in an equation and somehow know how fast the reaction will proceed. The equation only tells the amount of reactant and amount of product, but gives no info about how fast or slow reactant will give the product. Hence why this needs to be experimentally determined.
Here's an example with made up compounds with unknown elements:
10X -> 30Y + 15Z
all this can tell you, is that every 10 moles of compound X will give 30 moles of compound Y and 15 moles of compound Z. There is no info about how much time it will take: a few seconds, a few years, a few centuries. Only the amounts are given.
2A -> B + 3C
can you tell which of the two equations would be faster? No. Some may fall into the trap of thinking that since X has 10 moles and results in more moles of product that it must be faster. That may not be the case at all. A could take milliseconds while X could take decades, but vice versa could also be the case.
There is no way to determine rate from amount alone. All of these equations only give amounts. To get rate, take the compounds into a lab and experimentally determine how long the reactions take.
The purpose of this video, is to understand that the SLOWEST step (toll booth) determines the rate order. Also if the Slowest step happens to be the 2nd step or an intermediate, we know the intermediate used in the 2nd step was the product of the 1st, so we can sub the 1st steps reaction rate to get the intermediates concentration and continue from there.
Fantastic explanation!!
This is beyond helpful.
this video helped me a lot...thank you sir
Quick question: for the concentration for N202 and NO2, was the concentration to the power of 2 due to it being experimentally found at 2, or because there was two NO2 molecules reacting? Thanks :D
Nice vid!!
THANKYOU SO MUUUUUUUUUUUUUUUUUCH! I LEARNING TOO MUCH FROM YOUR VIDEO. XX.
Sincerely,
Your new subscribers~
Oh, yes it was helpful :) I really like your movies! Could you make one about apparent energy of activation of stepwise process? please :)
You know when my exercise physiology teacher (great professor) directs our class to your videos it's gotta be worth it. Videos like this definitely help me actually learn the information that I can't grasp from the textbook alone. Thank you for everything you're doing!!!and subbed finally lol
how do we know the intermediate whether will be incude in the equation?
you're awesome!! thank you:::)))
I literally have a test on this tomorrow!
In steady state approx rate of formation equals rate of disappearance of the intermediate, so why didn't you include k1=k-1+k2 as the steady state?
You are right, Ali. There is an issue when imposing the pseudo steady state hypothesis. In the video, Paul imposes an equilibrium hypothesis. This is only true at the thermodynamic equilibrium. To impose the pseudo steady state hypothesis correctly, you need to asume the net rate of generation and consumption of intermediaries is zero. In other words, you are right: k2 has to be part of the equation.
Question on K1 and K-1. At equilibrium would these be the same value? If so would they cancel out? Would the reverse rate ever occur due to the speed of the second step (fast step) and keep equilibrium, from being established which would make K-1 much smaller that K1? So many questions. Sorry. Great video!!!
Thank you!!!!!!!!
First saving me in AP Bio, now saving me again in AP Chem. *thank you*
Dude you are the Man!
How do you predict what the rate law is and how do we know what the slow step is?
RamAmandeep The rate law is the slowest step.
Hana Gabrielle Bidon No, the rate law includes the slow step plus every step before it, excluding the intermediates.
Well I am probably going to fail my chem final regardless if I know about this stuff. :'(
+RamAmandeep its prob too late. But they always tell u which one is the slowest.
Who needs college I learn everything here :)
perfect
why... there is equations with smallest k and he wrote (fast)equilibrium
Thans
ur a god
😘
Wait a minute. This is not about rate limiters...