Claisen Condensation Reaction Mechanism
HTML-код
- Опубликовано: 27 июл 2024
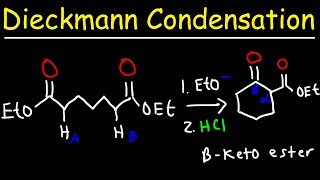
- This organic chemistry video tutorial provides a basic introduction into the claisen condensation reaction mechanism which involves the reaction of two esters and an alkoxide to produce a beta keto ester. This video contains plenty of examples and practice problems.
Subscribe:
/ @theorganicchemistrytutor
Access to Premium Videos:
/ mathsciencetutor
/ mathsciencetutoring
New Organic Chemistry Playlist
• Organic Chemistry - Ho...








Final Exams and Video Playlists: www.video-tutor.net/
Full-Length Math & Science Videos: www.patreon.com/mathsciencetutor/collections
this man has been carrying me through my major in college
What's your major?
This guy does better explanation than ph.d profesors, better examples than the online homework!
Thanks man. Been watching your vids for years now so again thank you.
Thank you for all your videos
great explanation sir!!!
This might be basic of me to say, but thanks again for all these great videos!
Bless you
How'd it go!?
bless you, gotten ,me through both Ochems!
Thanks for extras🙏🙏....it helped to understand better
Thank you once again.
this is a lifesaver
Great explanation Sir,
Would it be possible for you to make more videos on alkylation, acylation, adol reactions, beta elimination and related rxns, heterocyclic synthesis and reactivity
Pleaseee
They way you explain is very clear.
this man knows everything under the sun
Thanks!
Hello, can you please explain perkin condensation too?
Will the Ester part hydrolyse into alcohol if we have 2eq of hcl?
Thanks upload daily for neet ug 2019
Sir Why ethoxide ion not attack Alpha corbon in in the first reaction?
Awesome
Interesting
thanks
Nice
Can you make a video on perkin's Condensation,Please.
why does it attack the alpha-hydrogen only?
And also, why ethoxide is a good leaving group?
EtO- is weak base and also there is no other better leaving group than EtO-
why hydroxide can attack carbon directly but not when it was Eto? (Eto attacked alpha hydrogen first)
Because Eto is not sterically hindrance so it will not added to carbonyl group at first
Thanks
Can HCL be substituted with sulfuric acid?
@@asmaakram250 yes thank you, I was able to understand perfectly, a stronger acid can replace a weaker acid only in this reaction they are using HCl and I wanted to know if sulfuric acid a weaker acid can be used instead of HCl not the other way around. So for instance I would be using sulfuric acid to form the ketone.
I think it can be used instead only it would require a larger quantity of sulfuric acid over HCl I presume
yes, and it actually would be better
a stronger acid gives a better kinetic constant
but be careful with the concentration of the sulfuric, in my experience, going over 70% chars the reagents (basically reduces all organic compounds to elemental carbon, which is not great)
@@ivantimofeev2233 I appreciate your comment, thank you for your input.
The good thing is that the reaction takes place in an aqueous soln. and the sulfuric acid would be added slowly but I guess it would be best to pre-dilute the sulfuric acid before addition.
I actually ended up trying sulfuric acid and it worked, it was performed at mg scale just as proof of concept and it worked great. Took quite a lot of sulfuric acid, I was surprised.
bless you
why the LDA didn't react directly with carbonyl carbon at first but it react first with alpha hydrogen?
LDA isn't nucleophilic
Because Acid -base rxn happens at a very faster rate. Then Nu attack
Why does it needs to match?
🎉
Bless u
Lol