Pseudo Noble Gas Electron Configurations
HTML-код
- Опубликовано: 29 авг 2024
- We've learned about electron configurations, and also about how this plays into ionization, given that elements will tend to ionize in such a way so as to achieve noble gas electron configuration. But certain transition metals will seek to attain something slightly different, called pseudo noble gas electron configuration. Let's find out what that means!
Watch the whole General Chemistry playlist: bit.ly/ProfDave...
Study for the AP Chemistry exam with me: bit.ly/ProfDav...
Organic Chemistry Tutorials: bit.ly/ProfDave...
Biochemistry Tutorials: bit.ly/ProfDave...
Biology Tutorials: bit.ly/ProfDaveBio
Classical Physics Tutorials: bit.ly/ProfDave...
Modern Physics Tutorials: bit.ly/ProfDave...
Mathematics Tutorials: bit.ly/ProfDave...
EMAIL► ProfessorDaveExplains@gmail.com
PATREON► / professordaveexplains
Check out "Is This Wi-Fi Organic?", my book on disarming pseudoscience!
Amazon: amzn.to/2HtNpVH
Bookshop: bit.ly/39cKADM
Barnes and Noble: bit.ly/3pUjmrn
Book Depository: bit.ly/3aOVDlT



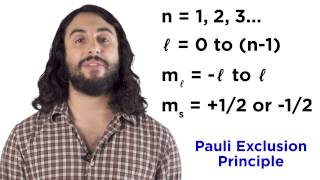





That's exactly what I wanted! You have clearly explained this. Actually I came here after I landed in a doubt in Fajan's rule which made me go crazy about what is pseudo noble gas configuration. I literally had no idea about that! Thanks anyway...
Exactly the same! Came from Fajans rule.
me too
Lol same
Same bhai
Same
I wanna binge-watch all of ur videos. They r just so good. Thank you, Professor Dave
THANK YOU SO MUCH PROFESSOR!!! Now I really have a clear understanding about this topic
Thank you sir ur vids r so understandable and simple ☺☺
Hello i am twelve years old and i was thinking that this lesson is a little bit hard, but later in your vedio it became more easy. Thank u🌹📖📚
This one is so effective. Thank you.
Thanks for the explanantion
Ur videos help lots of students
Thanks
Thank you sir
Hee this was interesting ánd usefull! I did not knew that this way of filling the orbitals even existed, so this is totally new knowledge for me (and for many others i think.) So thanx, again, for this lesson about this beautifull and interesting science topic.
thanks
Hi, I've been watching your General Chemistry videos 1 by 1 to to study Chemistry. Can you please explain why Ag has an electron configuration 5s1 4d10? I don't think you have mentioned this concept in any prior videos. You mentioned in the video at 2:20 this is according to the Aufbau principle, but the Aufbau principle would dictate the configuration to be 5s2 4d9 (fill up the lowest energy level first, and 5s has a lower energy level than 4d). I also re-watched your video on the Quantum numbers and Electron configuration but cannot find an explanation for this.
having a half full or completely full d subshell is stabilizing, enough to outweigh the energy difference between 5s and 4d for one electron, so it gets promoted
Can't Cr (4s1 3d5) also attain pseudo noble gas configuration as Cr^+1 (4s0 3d5) like Cu and Cd?
Thank you so much sir 🙏💖
Thnx sir from india
Thnx.
thanks professer
Long time long see. Professor dave
Thanks sir for clearing concept ❤️
what is pseudo noble gas
thanks professor 🙏🙏
PROFESSOR DAVE HELP!!! Why would n=4 for Ag be considered “full” at 18 e- when the 4th energy level can hold max 32 e-??!!
you said outermost but isnt it (n-1) shell of atom which after ionisation attains pseudo noble gas conf.
thanks prof
Thanks a lot 👍
Pls how did you do to have 6e on the reduction half équation🤔
I have a doubt...Can anyone help pls ????
Any cation or anion attain noble gas electronic configuration, so why every element cannot have pseudo noble gas configuration???
As Zn, Cd and silver have pseudo noble gas configuration when they are cation
Dave I love you
At last I got it.
science jesus saving me once again from the sins of a terrible school
Good
Can P block elements show Pseudo Inert Config ?
Yup heavier post transition elements ( p-block) exihibit this property in group three and four
@@lunam11 oh thanks
❤
Maaaaaaaaaannn urrr a leggeend
saved me
oh sorry about that Professor Dave
Well I searched the whole net for it.
jee maths it’s chemistry Jesus
1:57 It should be [Ar] not [Ne] but apparently no one cares lol
I searched it on Quora and chem stack exchange also.
I can understand your problem!
Jesus, is that you?
First !!!
U r❤️
Bro i gotta be honest you are pretty handsome
Can i translate your videos for arabs ????
yes that would be wonderful! you can submit them directly on any video and you can feel free to do this for any videos you like, it would be a tremendous help for me!
@@ProfessorDaveExplains are you intend that when i translat video send it to you to your channal ??
I iust can't understand
Yes, for any video, you click on the gear icon, click Subtitles/CC, then Add Subtitles/CC. You can choose a language and begin to add subtitles directly to the video. Then anyone in the world can watch the video with your subtitles! This is the easiest way to do it, as anyone can see it just by watching the video as it is. Any submissions are most appreciated! In fact, if you were willing to do them for many videos, like 50-100 or more, I would pay you.
do something cool for once
All my content is super cool, bro.