Catalysts | Kinetics | AP Chemistry | Khan Academy
HTML-код
- Опубликовано: 29 ноя 2014
- A catalyst is a substance that increases the rate of a chemical reaction without being consumed in the reaction. A catalyst works by providing a different pathway for the reaction, one that has a lower activation energy than the uncatalyzed pathway. This lower activation energy means that a larger fraction of collisions are successful at a given temperature, leading to an increased reaction rate. View more lessons or practice this subject at www.khanacademy.org/science/a...
Khan Academy is a nonprofit organization with the mission of providing a free, world-class education for anyone, anywhere. We offer quizzes, questions, instructional videos, and articles on a range of academic subjects, including math, biology, chemistry, physics, history, economics, finance, grammar, preschool learning, and more. We provide teachers with tools and data so they can help their students develop the skills, habits, and mindsets for success in school and beyond. Khan Academy has been translated into dozens of languages, and 15 million people around the globe learn on Khan Academy every month. As a 501(c)(3) nonprofit organization, we would love your help!
Donate or volunteer today! Donate here: www.khanacademy.org/donate?ut...
Volunteer here: www.khanacademy.org/contribut...








Best explanation I've seen so far on this... Good job!
Im gonna ace chemistry by watching your vids, thank you! Very well explained.
Thanks a lot, dear "Khan Academy" team. You are Great!
How do we know what catalysts create a faster response?
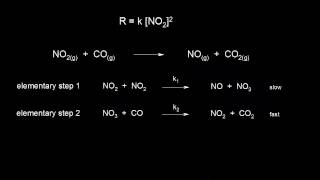
I think I read and watched about 20 explanations of how catalysts work. And this is the first one that mentions intermediate products being produced.
Without that, it's only telling what a catalyst does, but not an explanation of how it works.
Are there any reactions with more than one rate determining step? In which case how do you deal with those?
You add the times, if it takes 2 seconds for A to turn to B and 2 more seconds to B to turn to C it would be 4 seconds total. It's explained in the previous part of Kinetics, but usually the other steps happen so quickly compared to the rate determining step (10000x more) that when you round up it won't matter.
this is great, thanks
yes Anna is right catalyst alters the rate of rxn it can speed up rxn , Dec speed of rxn and can even kill the rxn
this was really helpful Mr. Khan .Nice job. Hats off to you........
Since I Is a catalyst how comes that it is counted for calculating molecularity ? Catalyst are not involved in calculating order and molecularity right ??
The bottom of Ea2 begins in the valley after Ea1. It does not begin at the low point for Ea1.
i need this video subtitle.
u give me pls ???
👍
but mr. khan anna and ashish are saying the right thing.
Por favor al castellano, gracias!!!
We learned that a catalyst alters the speed of a reaction not necessarily speeds it up.
a catalyst is generally used to speed up the reaction, what is does is provide an alternative pathway for the reaction
Exam on Wednesday fml
Does the catalyst size matter?
Yes the amount of catalysts does matter 4:25 , it increases the rate.